
Enzymes are proteins that catalyze most of the chemical reactions that take place in the body. They make it possible for chemical reactions to occur at neutral pH and body temperature. The chemical compound upon which the enzyme exerts its catalytic activity is called a substrate.
Proteolytic enzymes act on their natural substrates, proteins and peptides by hydrolyzing one or more peptide bond(s).
This process is usually highly specific in the sense that only peptide bonds adjacent to certain amino acids are cleaved.
Chromogenic substrates are peptides that react with proteolytic enzymes under the formation of color.
They are made synthetically and are designed to possess a selectivity similar to that of the natural substrate for the enzyme.
Attached to the peptide part of the chromogenic substrate is a chemical group which when released after the enzyme cleavage gives rise to color. The color change can be followed spectrophotometrically and is proportional to the proteolytic activity.
The chromogenic substrate technology was developed in the early 1970s, and has since then become a tool of substantial importance in basic research.
The majority of chromogenic substrate applications are found in various clinical fields. In particular they have been used to generate fundamental knowledge of the mechanisms regulating blood coagulation and fibrinolysis.
Furthermore, products based on chromogenic substrate technology have brought a new generation of diagnostics into the clinical laboratory.

Prothrombin, the natural substrate of Factor Xa, is cleaved by Factor Xa at two positions, each proceeded by the same four amino acid sequence. Factor Xa activity can be determined by the chromogenic substrate S-2222 which is composed of the same amino acids coupled to a chromophore.
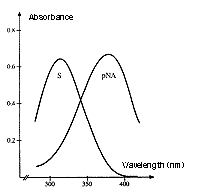
Absorption spectrum of a chromogenic pNA-containing substrate (S) and of pNA.
The hydrolysis of a peptide-pNA bond in the chromogenic substrates results in the release of pNA which in turn changes color. Thus the change in absorbance (ΔA/min) is directly proportional to the enzymatic activity.
The reaction is usually recorded at 405 nm.
